INTRODUCTION
Epithelial ovarian cancer (including ovarian, fallopian tube, and primary peritoneal cancers) is the deadliest gynecologic cancer, with a 5-year relative survival rate of only 50.8% in the US.1,2 Platinum-based chemotherapy can yield high response rates; however, most tumors will relapse and become resistant to platinum-based regimens.3 Standard-of-care chemotherapies for platinum-resistant ovarian cancer have shown limited clinical benefit, considerable toxicities, and historically poor prognosis with a substantial unmet need.4–7
Mirvetuximab soravtansine-gynx (ELAHERE®; AbbVie, Inc, North Chicago, IL) is an approved antibody-drug conjugate that targets folate receptor alpha, a biomarker frequently overexpressed in ovarian cancer and minimally expressed on normal tissue.8–12 The US Food and Drug Administration granted mirvetuximab soravtansine-gynx accelerated approval in November 2022 for folate receptor alpha-positive platinum-resistant ovarian, fallopian tube, or primary peritoneal cancer with 1–3 prior systemic treatment regimens based on results from the single-arm SORAYA trial (NCT04296890).13–15 The confirmatory, randomized, phase 3 MIRASOL trial (NCT04209855) demonstrated an unprecedented, significant survival advantage over chemotherapy and supported full approval in March 2024 followed by European Medicines Agency approval in November 2024.7,13,16,17
Anterior segment ocular adverse events have been documented for mirvetuximab soravtansine-gynx; prescribing information includes blurred vision or visual impairment (48%), keratopathy (36%), dry eye (27%), cataract (16%), photophobia (14%), eye pain (10%), and rarely, anterior uveitis (<1%).13 Ocular adverse events can be resolved through supportive care and dose modification when indicated.13 Effective mitigation and management strategies for mirvetuximab soravtansine-gynx–associated ocular adverse events, including prophylactic corticosteroid eye drop use, have been implemented in clinical trials and were previously described.18,19
Epithelial keratopathy, found with some classes of antibody-drug conjugates, has been previously described and given the term microcyst-like epithelial changes.20,21 While not fully elucidated, the pathogenesis of microcyst-like epithelial changes with mirvetuximab soravtansine-gynx may be due to apoptotic or antimitotic effects of the maytansinoid DM4 payload in the corneal epithelium.18,20 Microcyst-like epithelial changes have been associated with transient changes in corneal epithelial thickness profile and corneal topography, which may present as blurred vision or refractive shifts.20 Eye care professionals comanage these adverse events with oncologists by identifying the grade and communicating with the oncologist so that dosing can be held or modified, as indicated in Table 1.13 Because mirvetuximab soravtansine-gynx is continued until progression or toxicity, ocular adverse event management by eye care professionals is essential to maintain therapy.13
Herein, we describe a case with mild-to-moderate ocular adverse events that describe the clinical course of ocular adverse events experienced by patients receiving mirvetuximab soravtansine-gynx. In-depth information on the clinical course of more commonly observed ocular events with mirvetuximab soravtansine-gynx is provided, and the practical management of mirvetuximab soravtansine-gynx–related keratopathy is highlighted.22–24
A 56-year-old Black female with recurrent stage IIIC ovarian cancer (Müllerian carcinoma) had previously undergone radical excision of a pelvic tumor, total abdominal hysterectomy, bilateral salpingo-oophorectomy, colon resection, omentectomy, and ablation of a diaphragm tumor. The patient was treated with carboplatin, paclitaxel, pegfilgrastim, olaparib, and bevacizumab. At baseline ocular examination prior to starting mirvetuximab soravtansine-gynx, ocular history was positive for contact lens wear. Based on Pentacam imaging, corneal thickness at presentation was normal. Entering best-corrected distance visual acuity with moderate myopic correction was 20/20 + 2 in the right eye and 20/15 − 2 in the left eye. Vision improved to 20/15 in the right eye with refraction, and dilated examination findings were unremarkable aside from early lenticular changes. Loteprednol was prescribed, and cessation of contact lens was recommended per protocol to support ocular surface management.13
After 3 infusions of mirvetuximab soravtansine-gynx, the patient was seen for her first follow-up visit and reported blurred vision. Entering corrected acuities were 20/60 in the right eye and 20/40 in the left eye, and they improved to 20/20 in the right eye and 20/25 in the left eye with refraction, with a hyperopic shift (right eye > left eye). Anterior segment examination revealed mild superficial punctate keratitis with what appeared to be subepithelial pigmented microcyst-like epithelial changes in both eyes. Given that this was a low-grade adverse event, the patient continued mirvetuximab soravtansine-gynx treatment without dose modification.
Two weeks after the fourth infusion, the patient returned with worsening blurred vision. Entering acuities were 20/300 in the right eye and 20/400 in the left eye and did not improve with refraction. Anterior segment examination revealed moderate superficial punctate keratitis and pigmented microcyst-like epithelial changes in both eyes. Findings via macular optical coherence tomography were normal in both eyes. These findings were characterized as grade 3 keratopathy based on the reduction in best-corrected distance visual acuity and the Common Terminology Criteria for Adverse Events grading scale.25 The case was then discussed with the patient’s oncologist, who held mirvetuximab soravtansine-gynx dosing.
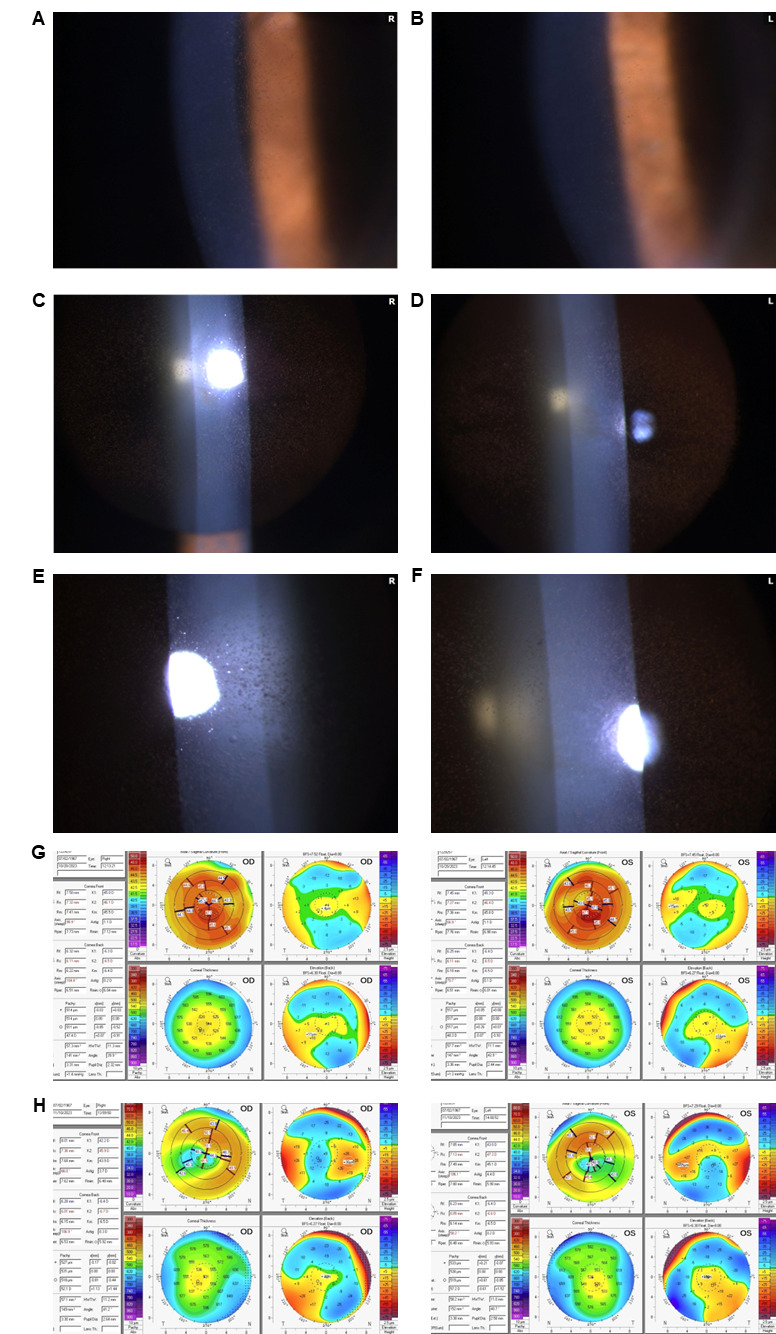
Three weeks later, the patient reported improved vision with entering distance acuities of 20/25 in the right eye and 20/30 in the left eye, which improved to 20/20 in the right eye with a mild change in refraction. There was no improvement in the left eye. The pigmented deposits had improved, and superficial punctate keratitis had nearly been resolved (Figure 1A and B). The patient resumed mirvetuximab soravtansine-gynx (fifth infusion) with a dose reduced by 1 level (5 mg/kg adjusted ideal body weight). The patient received the same dose 3 weeks later during her sixth infusion, and her vision continued to improve, with an “amazing” clinical response to treatment, per the oncologist.
After the sixth infusion, the patient reported that her vision without glasses had improved relative to baseline, to the level where she felt able to drive without glasses for the first time in her life. Uncorrected distance acuities were 20/25 right eye and 20/50 left eye, and there was a 4-diopter hyperopic shift from the patient’s previous myopic refraction, with no improvement in best-corrected distance visual acuity. However, at this lower dose, the patient experienced a recurrence of low-grade ocular adverse events after a few weeks. Slit lamp examination revealed superficial punctate keratitis and diffuse microcyst-like epithelial changes in both eyes, but no epithelial defect, nor iritis (Figure 1B–1F). Given the refractive shift, corneal topography taken 3 weeks prior (Figure 1G) was compared with that from this visit (Figure 1H). The corneal thickness profile had changed, with increases in overall corneal thickness corresponding to the appearance of microcyst-like epithelial changes. Anterior corneal topography revealed paracentral steepening decreasing mean keratometry. The oncologist was notified. Mirvetuximab soravtansine-gynx dose was then reduced to 4 mg/kg adjusted ideal body weight for infusions 7–10.
Following the tenth infusion, corneal findings had cleared, and best-corrected distance visual acuity improved to 20/20 in both eyes. The eleventh infusion of mirvetuximab soravtansine-gynx was given at a 1 dose level reduction from the starting dose (5 mg/kg adjusted ideal body weight).
DISCUSSION
This representative case demonstrates common ocular adverse events that patients may experience after mirvetuximab soravtansine-gynx treatment, including blurred vision associated with pigmented microcyst-like epithelial changes, and their clinical course. In this case, a hyperopic refractive shift was noted; similar refractive changes have been associated with other antibody-drug conjugates.20 Adding to previous literature reporting case studies of ocular events in patients receiving mirvetuximab soravtansine-gynx, which is limited to 3 reports,22–24 the current case study reported detailed information on hyperopic refractive changes over an extended period with mirvetuximab soravtansine-gynx. The case offers a real-world example of the collaborative management of more commonly observed ocular events with mirvetuximab soravtansine-gynx.
In an integrated safety analysis of 682 patients from 4 mirvetuximab soravtansine-gynx monotherapy clinical trials, grade 3 ocular adverse events occurred in 11% of patients, and grade 4 ocular adverse events occurred in 0.3% of patients.26 For those with ocular adverse events, 53% had complete resolution, 38% had partial improvement, and 1% permanently discontinued mirvetuximab soravtansine-gynx due to ocular adverse events.26 However, for a subset of patients, including the patient described in the present study, symptoms can be higher grade and can require more active intervention. Of note, the most common ocular adverse events, blurred vision, and keratopathy (grouped terms), typically occurred at 5.9 weeks (range, 0.1–56.3) and 6.7 weeks (range, 0.1–8.1), respectively,26 which aligns with what was observed in the case reported here.
In this report, ocular surface inflammation was not a notable initial finding when blurred vision occurred. Consequently, additional topical or systemic steroid therapy was not warranted to manage microcyst-like epithelial changes. In other cases seen at our institution, patients were able to continue mirvetuximab soravtansine-gynx following the first occurrence of blurred vision and reported improvement at their next ophthalmic examination 3 weeks later without topical or systemic steroids. In one case, microcyst-like epithelial changes were managed without dose modification. These findings support microcyst-like epithelial changes as a manifestation of initial payload toxicity.19
This report illustrates the eye care professional’s crucial role in determining dose adjustments of mirvetuximab soravtansine-gynx. The patient described here experienced epithelial keratopathy with a grade 3 reduction in best-corrected distance visual acuity after 4 treatment cycles, necessitating a dose hold and reduction. Keratopathy improved after the modification, with no apparent cancer progression during the follow-up period reported here. This finding corresponds to results from MIRASOL, which showed a significant survival benefit with mirvetuximab soravtansine-gynx, even though 20% of participants received dose reductions and 54% received dose delays/holds due to ocular adverse events.16,27 This report highlights that ocular events may fluctuate over time and with mirvetuximab soravtansine-gynx dosing. Vigilant monitoring is essential, as patients may be asymptomatic or exhibit intermittent symptoms requiring coordination with oncology.18 Importantly, a prior study demonstrated that patients maintain clinical response with mirvetuximab soravtansine-gynx dose modifications and reductions.28
The clinical appearance of microcyst-like epithelial changes may be variable. The patient in our report, who has highly pigmented skin, had microcystic changes that presented with pigment deposition. One historical report found hyperreflective material on confocal microscopy in patients receiving a different antibody-drug conjugate (belantamab mafodotin),21 a finding that may account for the differential appearance of microcystic changes across patients. The implication of varying appearances of microcyst-like epithelial changes among patients is unclear, but this finding may represent corneal epithelial cells arrested in different stages of proliferative phase and warrants further investigation.
CONCLUSION
Mirvetuximab soravtansine-gynx is the first treatment for platinum-resistant ovarian cancer to demonstrate a survival advantage over single-agent chemotherapy.16 Unlike prior regimens, it is associated with new ocular toxicities, necessitating collaboration with eye care professionals to provide patient counseling, management of ocular surface disorders, and communication with oncologists to support early treatment modifications. This case demonstrates that modification of mirvetuximab soravtansine-gynx dosage can be an effective strategy to mitigate ocular adverse events and that most events resolve on their own or after dose adjustment. Further, adherence to both steroid and lubricating eye drops as prophylactic measures appears to be effective in diminishing the incidence of ocular adverse events and ocular adverse event-related discontinuations. Although vision changes can be distressing, eye care professionals can reassure patients that mild ocular adverse events typically occur within the first month and are manageable with appropriate treatment adjustments. For patients undergoing treatment with mirvetuximab soravtansine-gynx, eye care professionals are an indispensable part of the care team. Eye care professionals should communicate frequently with oncologists to ensure that ocular adverse events are monitored and effectively managed, so that together, they can ensure the highest level of care for their shared patients.
TAKE HOME POINTS
-
Ocular adverse events are common in clinical trials of mirvetuximab soravtansine-gynx, an antibody-drug conjugate for ovarian cancers.
-
A representative case of a patient treated with mirvetuximab soravtansine-gynx is presented here.
-
Ocular adverse events were resolved with dose delay or dose adjustment of mirvetuximab soravtansine-gynx.
Patient Consent
Consent to publish the case report was not obtained; no identifiable health information was included in this case report.
Human Rights
This case report was conducted in accordance with recognized ethical principles and adhered to all applicable regulations governing the protection of human subjects, including the tenets of the Declaration of Helsinki. All identifying information has been removed to protect patient privacy.
Conflicts of Interest
Kristen P. Lamoreau reports receiving honoraria from ImmunoGen, Inc. for focus group participation and involvement in an educational video. Jae Young You reports receiving honoraria from ImmunoGen, Inc. for participation in an advisory board.
Funding Sources
This study was supported by ImmunoGen, Inc (now a part of AbbVie, Inc).
Data Availability
The clinical data for the patient described in this case report are no longer available. As this manuscript reports on a single patient encounter and no additional datasets were generated or retained, no supporting data are available for sharing.
Commercial Sponsorship
Development of this manuscript was funded by ImmunoGen, Inc (now a part of AbbVie, Inc). The sponsor had no role in the clinical management of the patient but provided support for manuscript preparation.
Artificial Intelligence (AI) Use
No artificial intelligence tools were used in the development of this manuscript or the processing of data reported within.
ACKNOWLEDGMENTS
We thank the patients and their families. Medical writing assistance was provided by Gloria Marino Bravante, PhD, Amanda Agazio, PhD, CMPP, and Kelsey Pinckard Schaefers, PhD, of Precision AQ (Bethesda, MD, USA) and funded by ImmunoGen, Inc (now a part of AbbVie, Inc).