INTRODUCTION
Demodex mite infestation (demodicosis) of the eyelids occurs with 2 species of mites: Demodex brevis, which primarily inhabits the meibomian glands, and Demodex folliculorum, which typically resides in the eyelash follicles.1 The global prevalence of infestation on the eyelashes has been estimated at 33%, with an increase in older adults and eyecare patients.2 Although these mites are considered a normal part of the skin flora and may present without symptoms,3 growing evidence has linked demodicosis with blepharitis, chalazion, rosacea, and skin inflammation.4–6
A pathognomonic sign of Demodex mites is cylindrical, sleeve-like coatings at the base of the eyelashes,7 referred to in the literature as collarettes, cylindrical dandruff, or sleeves. The number of collarettes per eyelid serves as a surrogate metric of mite density and can be graded on the following scale: grade 0 is 0 to 2 collarettes, grade 1 is 3 to 10 collarettes, grade 2 is >10 collarettes to <1/3 of the eyelashes have collarettes, grade 3 is ≥1/3 to <2/3 of the eyelashes have collarettes, and grade 4 is ≥2/3 of the eyelashes have collarettes.8
Various treatments for Demodex-associated blepharitis have been used with varying degrees of success. Routine face cleansing and baby shampoo eyelid scrubs are ineffective at eliminating the mites.9 Hypochlorous acid solution is not demodicidal.10 Tea tree oil exhibits diminished demodicidal activity at concentrations below 50%,11,12 while concentrations of 50% and above can cause significant ocular and skin irritation.13 Terpenen-4-ol, an active component of tea tree oil, can control mite populations but is insufficient for eradication and has been shown in-vitro to be toxic at low concentrations to human meibomian gland epithelial cells.10,14 Various other essential oils, Manuka honey, and okra-based eyelid wipes, show promise but have not been extensively studied.4,15 Intense pulsed light does not result in a statistically significant decrease in mite counts.16 In 2023, 0.25% ophthalmic lotilaner (XDEMVY, Tarsus Pharmaceuticals, Irvine, CA, USA) was approved in the United States for the treatment of Demodex blepharitis17; however, access may be cost prohibitive.18
In contrast, multiple studies have demonstrated the effectiveness of off-label, topical 1% ivermectin cream in treating Demodex-associated blepharitis.19–22 In 2020, nonprescription 0.5% ivermectin lotion marketed as a head lice treatment became available in the United States.17 Given the general accessibility and affordability of nonprescription medications, this development raises the possibility that 0.5% ivermectin lotion could be a convenient and novel treatment option for Demodex-associated blepharitis.
CASE REPORTS
Each patient in this case series underwent 2 in-office applications of 0.5% ivermectin lotion performed 2 weeks apart for symptomatic Demodex-associated blepharitis. Slit-lamp biomicroscopy was performed and the eyelids were photographed before each ivermectin application and at final follow-up. Treatment was delivered using a modified Helm protocol.19 Initially, a drop of 0.5% proparacaine hydrochloride was instilled into each eye. The exam chair was reclined, and 0.5% ivermectin lotion was applied to the base of the upper and lower lashes with a cotton-tipped applicator, taking care to keep the lotion off the ocular surface. After saturating the upper and lower eyelashes, the lotion was applied to the surrounding eyelid skin and eyebrows. It was left in place for 10 minutes and then removed with a sterile-saline soaked eye pad. A small amount of residual lotion was purposefully left at the base of the lashes. Finally, as a precaution, the ocular surface was rinsed with 0.4 mL of preservative-free artificial tears before the patient was discharged.
The details of each case are described below.
Case 1
A 70-year-old White woman presented with intermittent eyelid itching and constant blurred vision for the past year. Carboxymethylcellulose artificial tears had not improved her symptoms. She also noted persistent debris at the base of her eyelashes that had been unresponsive to micellar eyelid wipes. The patient’s ocular history was significant for epithelial basement membrane dystrophy without corneal erosions. She had been prescribed sodium chloride ophthalmic solution and ointment for this in the past but had stopped after perceiving no effect. Her medical history was significant for rosacea, anxiety, osteoarthritis, vitamin D deficiency, and gastroesophageal reflux disease. For these, she was taking alprazolam, cholecalciferol, and omeprazole, but did not take medication for rosacea. She was allergic or had suffered adverse reactions to penicillin, etodolac, and oral metronidazole.
On examination, best corrected visual acuity was 20/25-2 in each eye. Intraocular pressure was 16 mm Hg in the right eye and left eye by rebound tonometry. Pupil testing, extraocular motility, and confrontation visual fields were normal in both eyes. There were mild, fine telangiectasias on the cheeks. Slit-lamp biomicroscopy revealed grade 3 collarettes on the upper eyelashes and minimally raised, thin curvilinear irregularities of the corneal epithelium within the visual axes without corneal scarring in both eyes. The conjunctivas were white and quiet, and the anterior segment was otherwise unremarkable in each eye. Dilated fundus examination revealed bilateral posterior vitreous detachments but was otherwise normal.
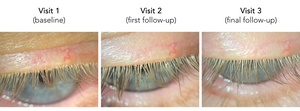
The patient was diagnosed with Demodex-associated blepharitis and mild corneal epithelial basement membrane dystrophy in both eyes; her itching was attributed to the Demodex-associated blepharitis and her blurred vision to the epithelial basement membrane dystrophy. She declined any corneal procedures or further treatment for the corneal dystrophy. For management of the Demodex-associated blepharitis, she initially agreed to an in-office treatment with prescription 1% ivermectin cream. However, her health insurance declined coverage for this medication and therefore, after careful consideration and informed consent, she declined an at-home regimen of 0.5% ivermectin lotion and proceeded with in-office administration of the medication instead (Figure 1).
At the patient’s 2-week follow-up, she reported that her eyelid itching had been resolved. She denied any significant discomfort during or following the procedure but did note a temporary increase in itching on the second and third days following the treatment, which subsided. The patient also reported continued daily eyelid hygiene with micellar eyelid wipes and stable vision. On repeat examination, her corneal appearance remained unchanged. However, grade 1 collarettes were observed on the upper eyelashes and thus a second in-office treatment with 0.5% ivermectin lotion was administered (Figure 1). The patient returned 2 months later with symptoms of mild eyelid dryness but no itching and had stable vision. She expressed satisfaction with the results. Examination revealed grade 0 collarettes on the eyelids while her other anterior segments findings remained stable (Figure 1).
Case 2
A 73-year-old White man presented with burning and itching of the eyelids that had been unresponsive to eyelid scrubs, cool compresses, and hypochlorous acid spray eyelid cleanser. The patient’s ocular history was significant for 5 prior episodes of Bell’s palsy that had affected both sides of his face. He also had brow ptosis and dermatochalasis for which an oculoplastic surgeon had deferred surgery. His medical history was significant for diabetes mellitus type 2, hypertension, hypercholesterolemia, cerebrovascular accident, myocardial infarction, iron deficiency anemia, benign prostatic hyperplasia, and gastroesophageal reflux disease. He was taking the following medications and supplements: metformin, carvedilol, losartan, atorvastatin, aspirin, clopidogrel bisulfate, ferrous gluconate, ascorbic acid, docusate, tamsulosin, and pantoprazole. He was allergic or had suffered an adverse reaction to codeine.
On examination, best corrected visual acuity was 20/25-2 in each eye. Intraocular pressure was 12 mm Hg in the right eye and left eye by rebound tonometry. Pupil testing, extraocular motility, and confrontation visual fields were normal in both eyes. There were trace to mild fine telangiectasias on the cheeks, a moderate brow ptosis, and moderate dermatochalasis of the eyelids. Orbicularis function was intact and there was no lagophthalmos. Slit-lamp biomicroscopy revealed grade 3 collarettes on the upper eyelashes of each eye; the anterior segment was otherwise unremarkable. A review of records revealed no pathology on a recent dilated funduscopic examination.
The patient was diagnosed with Demodex-associated blepharitis. Treatment with 0.25% ophthalmic lotilaner was deferred due to insurance-mandated step-therapy requirements. The patient declined an at-home regimen of 0.5% ivermectin lotion and instead proceeded with in-office administration of the medication, which was performed without incident (Figure 2).
At the patient’s 2-week follow-up, he reported a 60% to 65% improvement in his burning and itching symptoms. He had experienced mild discomfort for 30 minutes after the initial treatment while on his way home, which he described as being similar to having soap in his eyes. On repeat examination, grade 1 collarettes were noted on the upper eyelashes while his other anterior segments findings remained unchanged (Figure 2). A second in-office ivermectin treatment was administered without incident. The patient returned 1 month later and reported that his eyes felt comfortable. He had not been performing any ongoing eye treatments at home. As before, he had experienced mild discomfort for about 30 minutes after the second treatment; this was ameliorated by wiping his eyelids with a cloth. Despite the initial discomfort, he expressed satisfaction with the results. Examination revealed grade 0 collarettes on the eyelids but otherwise stable anterior segment findings (Figure 2).
Case 3
A 57-year-old White man was referred for treatment of his Demodex-associated blepharitis after his health insurance had denied coverage of 0.25% ophthalmic lotilaner. He reported an intermittent foreign body sensation in the right eye and a persistent feeling of thickness and tightness in the right lower eyelid. Carboxymethylcellulose artificial tears had not improved his symptoms. The patient’s ocular history was significant for Demodex-associated blepharitis, diagnosed several months before his referral as well as attempted laser-assisted in situ keratomileusis in the right eye over a decade prior. His medical history was significant for allergic rhinitis, asthma, osteoarthritis, erectile dysfunction, and gastroesophageal reflux disease. He was taking the following medications: fexofenadine, montelukast, albuterol, mometasone, meloxicam, sildenafil citrate, and omeprazole. He reported no drug allergies.
On examination, best corrected visual acuity was 20/25+1 in the right eye and 20/20 in the left eye. Intraocular pressure was 10 mm Hg in the right eye and 12 mm Hg in the left eye by rebound tonometry. Pupil testing, extraocular motility, and confrontation visual fields were normal in both eyes. There were mild, fine telangiectasias on the cheeks. Slit-lamp biomicroscopy revealed grade 4 collarettes on the upper and lower eyelashes and saponification of the tear film on the lower eyelid margins of each eye. There was a small chalazion in the right lower eyelid visible on eyelid eversion. The right cornea had an irregular paracentral scar consistent with a healed, laser-assisted in situ keratomileusis flap buttonhole and the left cornea was clear. The anterior segment of both eyes was otherwise unremarkable. A review of records indicated no pathology on recent funduscopic examination.
The patient was diagnosed with meibomian gland dysfunction, a chalazion, and his Demodex-associated blepharitis was confirmed. The sensation of eyelid tightness was attributed to the chalazion while the foreign body sensation was attributed to a potential combination of the aforementioned conditions. The patient was advised to perform warm compresses for the meibomian gland dysfunction and chalazion. After careful consideration and informed consent, he proceeded with an in-office treatment of 0.5% ivermectin lotion for the Demodex-associated blepharitis (Figure 3).
At the patient’s 2-week follow-up, he reported that the foreign body sensation had resolved but the feeling of tightness in the right lower lid remained unchanged. He denied any discomfort during or following the initial procedure. On repeat examination, grade 2 collarettes were noted on the right upper eyelashes only and there were no collarettes on right lower eyelashes or on the left eyelashes (Figure 3). The chalazion persisted and his corneal appearance was stable. Given the unilateral collarettes, the patient elected to proceed with repeat treatment on the right side only. He returned in 1 month and reported that the foreign body sensation had not returned but that his right lower eyelid still felt tight. Examination revealed the chalazion in the right lower eyelid remained unchanged and showed grade 0 collarettes on the eyelids (Figure 3). His other anterior segment findings were stable. The patient resumed care with his regular eyecare provider as planned.
DISCUSSION
Ivermectin is a macrocyclic lactone derivative isolated from Streptomyces avermitilis.23 Since its discovery in the 1970s, it has been used as a broad-spectrum antiparasitic agent in both human and veterinary medicine. It acts by binding to invertebrate-specific glutamate-gated chloride ion channels, potentiating channel activity and causing hyperpolarization of parasites’ neurons and muscles, thereby paralyzing and killing them.23 The use of oral ivermectin, topical ivermectin, and ivermectin combined with metronidazole has been studied for the treatment of Demodex-associated blepharitis.4,24 Although multiple papers have demonstrated the efficacy of prescription 1% ivermectin cream in the treatment of Demodex-associated blepharitis,19–22 a search of the PubMed database identified no previous reports describing the use of nonprescription 0.5% ivermectin lotion for this condition.
Patients in this case series demonstrated a reduction in collarettes of at least 2 grades after the first administration of ivermectin, and all patients achieved grade 0 collarettes after the second administration. The marked improvement observed with only 1 or 2 ivermectin applications suggests that prolonged daily or weekly treatment over months, as described by other authors,20–22 is unnecessary. The improvement after limited ivermectin applications discussed here replicates the findings of Helm and Rivera Salazar et al.19,25 Therapeutic effect persisted throughout the limited 1 to 2-month follow-up period; however, modest collarette recurrence has been reported 6 to 16 months after topical ivermectin treatment.19
Symptoms attributable to Demodex-associated blepharitis improved in all patients after a single administration of 0.5% ivermectin. The finding that residual collarettes remained after the first administration of ivermectin while symptoms improved implies that complete eradication of Demodex mites is not necessary to achieve symptomatic relief. In fact, it has been suggested that the therapeutic objective in Demodex management should be to restore ocular ecological balance rather than to achieve complete elimination of the organism.26 Because itching is a primary symptom of Demodex-associated blepharitis and is not assessed by the Ocular Surface Disease Index,27 that questionnaire was not used to quantify patient symptoms in this case series.
Several considerations influenced the treatment protocol. The fact that the lifecycle of Demodex folliculorum spans 2 to 3 weeks informed the decision to administer 2 applications of 0.5% ivermectin separated 2 weeks apart.26 Previous studies have used various durations of 1% ivermectin cream contact to kill the mites: Helm used 10 minutes,19 Choi et al used 15 minutes,20 and Rivera Salazar et al used upwards of 18 minutes.25 Notably, 10 minutes is the treatment time recommended by the 0.5% ivermectin lotion package insert for the eradication of head lice.28 The 10-minute contact time used in this case series proved sufficient.
Application of ivermectin lotion to the eyelids and adnexa was well tolerated by the patients. This observation is consistent with findings by Valvecchia et al, who reported occasional discomfort in only 0.6% of participants (14 of 2157) treated with 1% topical ivermectin once daily for 2 months.22 In case 1, the patient experienced an increase in eyelid itching on day 2 and day 3 following the procedure. Given that this symptom did not recur following a second application of ivermectin, it was hypothesized that the initial exacerbation resulted from an inflammatory response to an extensive Demodex mite die-off. Notably, an increase in dry eye symptoms several days after treatment was reported by Helm.19 In case 2, the patient reported that it had felt like there was soap in his eyes for 30 minutes after each application of ivermectin lotion. Because this symptom improved by wiping the eyelids with a cloth, it was hypothesized that residual ivermectin lotion at the base of the eyelashes following the procedure was migrating onto the patient’s ocular surface.
The corneal status immediately following ivermectin lotion removal was not assessed. However, ocular surface health in patients treated with topical 1% ivermectin cream has been evaluated. Choi et al reported improvement in Oxford staining scores across 51 patients treated with 1% ivermectin cream applied for 15 minutes once weekly in conjunction with daily eyelid hygiene over an average of 15 weeks.20 Similarly, Smith et al reported improvement in fluorescein staining scores across 70 patients treated with 1% ivermectin cream alone, applied daily for 3 months and assessed at the 3-month mark.21 These results suggest that any potential ocular surface toxicity induced by applying 1% ivermectin cream to the eyelids is transient at worst. Although these findings are likely applicable to 0.5% ivermectin lotion, further investigation is warranted.
Although ivermectin lotion was applied directly to patients by the author in this case series, prior studies have demonstrated that patient-administered, at-home application of topical ivermectin to the eyelids is safe and effective.20–22,24 In this case series, however, patients generally preferred doctor-administered, in-office treatment when presented the option. The reasons for this preference were not elicited, but potential contributing factors in the general population include dexterity limitations, visual impairment, concern about ocular exposure, and caregiver constraints. Nevertheless, at-home therapy offers potential health care cost savings.
Finally, although much is known about Demodex infestation of the eyelids, significant questions remain.
It is unclear to what degree Demodex mites can cause blepharitis, rosacea, and skin inflammation, or whether they simply co-occur in these conditions. If they are pathogenic, the underlying mechanisms have yet to be elucidated, and the optimal treatment strategies remain uncertain. Essentially, the extent to which Demodex mites function as harmless commensals versus pathogenic organisms, and the conditions under which they may transition between these roles, remains unknown. Importantly, there is also no consensus that Demodex mites directly cause dry eye disease or meibomian gland dysfunction.4 Accordingly, eye care providers must be judicious when recommending treatment for Demodex mites.
CONCLUSION
This case series suggests that 2 in-office applications of topical 0.5% ivermectin lotion, administered 2 weeks apart, are an effective treatment to reduce the eyelash collarettes associated with Demodex infestation. Although multiple studies have demonstrated the efficacy of prescription 1% ivermectin cream in the treatment of Demodex-associated blepharitis, nonprescription 0.5% ivermectin lotion for this indication has not been previously reported. Because nonprescription medications are typically more accessible and affordable than prescription-only therapies, these findings may inform future research directions and treatment approaches for patients with symptomatic Demodex-associated blepharitis.
TAKE HOME POINTS
-
In-office application of topical 0.5% ivermectin lotion is an effective treatment to reduce the eyelash collarettes associated with Demodex infestation.
-
0.5% ivermectin lotion, sold as head lice shampoo, is available without a prescription in the United States.
-
There remains no consensus that Demodex mites directly cause dry eye disease or meibomian gland dysfunction.
Patient consent
No identifiable health information was included in this case series.
Human Rights
Case series reporting 3 or fewer patients do not require institutional review board approval.
Conflict of Interest
The author declares no conflicts of interest.
Funding Sources
The author declares no funding sources.
Data Availability
Data sharing is not applicable to this article as no new data were created or analyzed in this case series.
Artificial Intelligence Use
No artificial intelligence was used in the preparation of this manuscript.
Credit for photo on main article page: Christopher Lievens, OD, MS, PhD, FNAP, FAAO