Atopic dermatitis is a chronic, pruritic, eczematous disease that frequently starts in childhood and can continue throughout life with periods of remission and flaring. It develops because of a complex interrelationship of environmental, immunological, genetic, and pharmacological factors. Exacerbations of atopic dermatitis can occur with infections, psychological stress, climate/seasonal changes, allergens, and irritants. This presents as inflammation of the skin involving hypersensitivity, immunoglobulin E sensitization, and T-helper cell 2 activation. This results in severe pruritus and xerosis.1,2 Atopic dermatitis mostly affects children but does occur in 5% of adults.3–5 Atopic keratoconjunctivitis, an ocular manifestation of atopic dermatitis, is characterized by severe ocular pruritus and eyelid dermatitis.6 Atopic keratoconjunctivitis involves both types 1 and 4 hypersensitivity reactions, causing inflammation of the periocular skin, conjunctiva, and cornea.6 Conjunctival scarring, limbal stem cell deficiency, and corneal ulceration are severe complications that may lead to vision loss.6,7 To improve the quality of life in patients experiencing atopic dermatitis, systemic immunosuppression is often necessary. Dupilumab (DUPIXENT, Regeneron & Sanofi, USA) was the first biologic that was approved for the treatment of atopic dermatitis in 2017. However, this highly effective treatment is associated with an increase incidence of conjunctivitis, blepharitis, keratitis, and eye pruritis, which present similarly to atopic keratoconjunctivitis. These side effects are known as dupilumab-associated ocular surface disease. Although there are ocular surface complications associated with atopic disease, corneal ulceration has rarely reported to be directly associated with the use of dupilumab.3,7–11 However, the management and treatment is similar between atopic keratoconjunctivitis and dupilumab-associated ocular surface disease. No identifiable health information was included in this case report.
CASE REPORT
Dermatology referred a 21-year-old multiracial man for ocular inflammation. He had a long-term history of atopic eyelid dermatitis with lower-lid ectropion. He reported no surgical eye procedures. The patient had noted increased redness, injection, and foreign body sensation in both eyes for the past 3 months.
Medical history was significant for atopic dermatitis since childhood, hyperimmunoglobulin E syndrome, and inflammatory bowel disease. Systemic medications included vedolizumab (ENTYVIO, Takeda Pharmaceuticals, USA) for 2 years and topical tacrolimus 0.03% for skin eruptions. He experienced failed treatment with other therapies, including infliximab, certolizumab, and adalimumab. Current ocular medications included 0.3% tobramycin/0.1% dexamethasone ointment applied to the eyelids twice a day where the skin eruptions persisted. He reported using fluorometholone 0.1% drops in the past, which were prescribed by his primary care physician.
The examination revealed uncorrected visual acuities of 20/20 in the right eye and 20/25 in the left eye. The pupils were round and reactive to both light and near targets. There was no afferent pupillary defect noted. Extraocular motilities were full, and confrontation visual fields did not demonstrate any central or peripheral depressions or defects in either eye.
The external examination demonstrated patchy, eruptive lesions of the forehead and periorbital dermis, both eyes. Biomicroscopic examination showed thickened inferior eyelids with excoriation. Mild scurfs were present. There was 1+ injection of the bulbar conjunctiva and noted 1+ giant papillae on upper-lid eversion of the palpebral conjunctiva, both eyes. Scant mucoid discharge was seen in the fornixes. The corneal surface was normal in appearance, without significant punctate epithelial erosions in both eyes. The anterior chambers did not demonstrate cells or an inflammatory process in either eye. The crystalline lens did not show any cataract formation in either eye. Tonometry was 16 mm Hg in the right eye and 15 mm Hg in the left eye.
The vitreous bodies were intact. The optic disc had 0.25/0.25 cupping with symmetrical neuroretinal rim tissue in both eyes. The maculae were normal, and the peripheral retina tissue did not demonstrate any defects in either eye.
The tentative diagnoses at this time were atopic blepharoconjunctivitis and periocular dermatitis. Management included discontinuing the 0.3% tobramycin/0.1% dexamethasone ointment and replacing it with tacrolimus 0.03% ointment twice a day for both eyes. Preservative-free artificial tears were to be used, as well as humidification of his environment. Use of olopatadine 0.7% once a day for both eyes was also recommended rather than a topical corticosteroid since it was felt a mast-cell stabilizing was thought to more useful for long-term maintenance. The patient was scheduled to come back in 6 months for a comprehensive examination.
Two months after the consultation, the patient called and said that he was in “excruciating” pain. He reported that he was using more than eight vials of preservative-free artificial tears a day with no relief. He had self-discontinued the tacrolimus ointment since the skin lesions had improved. Of significance, he had begun dupilumab treatment for his atopic dermatitis 6 weeks prior to his phone call. He received his second dose 3 days prior to his symptoms increasing.
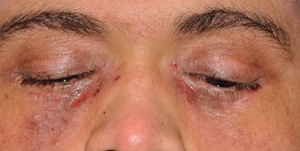
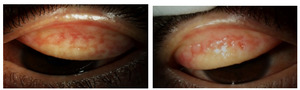
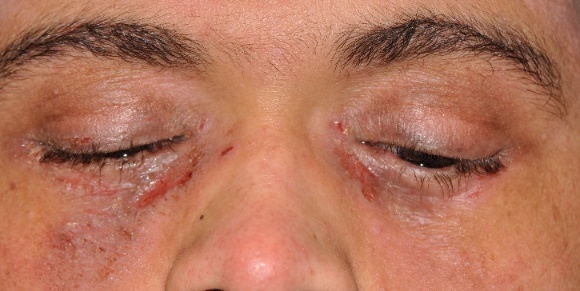
Pertinent examination findings showed 2+ scaling and eruptions of the skin on the inferior periorbital region of both sides (Figure 1). Thickening and ptosis of the upper lids was evident. There was mild ectropion of the lower eyelids, both eyes, and lagophthalmos with inferior exposure of the ocular surface, left eye (Figure 2). Biomicroscopy demonstrated 2+ diffuse conjunctival bulbar injection without ciliary flush, both eyes. On upper lid eversion, 3+ giant papillae were evident (Figure 3). There was 2+ inferior corneal punctate erosions. The anterior chamber was quiet.
The history and signs indicated a tentative diagnosis of dupilumab-associated ocular surface disease. The patient was placed on prednisolone acetate 1% 4 times a day; olopatadine 0.7% twice a day, both eyes; preservative-free artificial tear drops every 4 hours, and nighttime ointment. He was instructed to restart the tacrolimus 0.03% ointment twice a day to the facial eruptions. A discussion with his dermatologist resulted with the discontinuation of further dupilumab injections. He was scheduled for a 1-week follow-up.
The patient did not return for his scheduled follow-up visit and presented to urgent care 3 weeks after his last examination, with the complaint of blurred vision, pain, and eye redness in the right eye for several days. He did not comply with treatment recommendations and self-discontinued with his topical medications except for artificial tears. He had not received another dupilumab injection. The pain was rated at 10 out of 10. He was diagnosed with allergic conjunctivitis and given olopatadine 0.1% drops 4 times a day, both eyes, along with use of preservative-free artificial tears. Urgent care also gave oral prednisone for the eczema. He also tried cool compresses, oral loratadine, and fluticasone propionate nasal spray without relief. He was scheduled to be seen in the vision services department the next day.
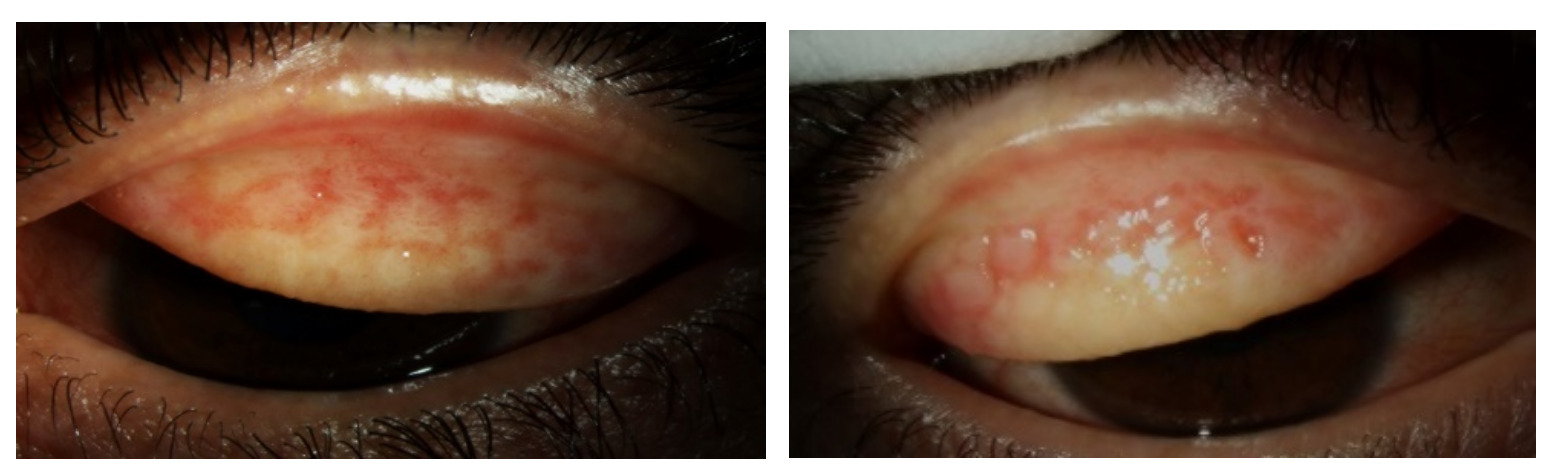
On ophthalmic examination the next day, uncorrected visual acuity was 20/80 right eye and 20/40 left eye. There was moderate photophobia present. The eyelid dermis was stable without new eruptions or erythema. Biomicroscopy demonstrated increased chemosis and injection of the bulbar conjunctiva right eye greater than the left eye. The right cornea showed a large round peripheral defect 3 mm in size with several nodular infiltrates centrally and an immune ring about 6 mm (Figure 4). There was diffuse 3+ staining sodium fluorescein of the defect. There was no mucous discharge. The anterior chamber showed a mild inflammatory reaction without hypopyon. Multiple ESwab (Copan Diagnostics) cultures of the corneal surface, the upper and lower palpebral conjunctiva, and surrounding lid margins were performed. A corneal scaping was also performed. The samples were sent to the hospital laboratory for bacterial, viral, and fungal analysis. A real-time polymerase chain reaction assay was also ordered. The left conjunctiva and cornea were unchanged from previous examination. Since the symptoms and appearance were suggestive of infectious keratitis, the patient was treated with moxifloxacin 0.5% ophthalmic solution every 4 hours and fortified tobramycin 14 mg/mL drops every 4 hours in alternating fashion until the results of the cultures were known. The patient also was given cyclopentolate 1% twice a day in the right eye. The patient was instructed to continue to use preservative-free artificial tears in both eyes. He was to resume use of the tacrolimus 0.03% ointment to the periocular lid tissue, right eye.
The patient was seen 2 days later and reported mild improvement in comfort and vision. He reported compliance with the moxifloxacin, tobramycin, and cyclopentolate. Examination showed improved uncorrected visual acuity to 20/40 in the right eye and 20/30 in the left eye. Slit lamp revealed a smaller defect about 2 mm in size. The immune ring and infiltrates were still present. Conjunctival injection was reduced, and the anterior chamber reaction was limited to trace cells. The bacterial and fungal cultures showed no growth at 24 and 48 hours. The polymerase chain reaction assay was pending.
Since there was no pathogen growth and the defect was smaller, tobramycin was replaced with 0.3% tobramycin/0.1% dexamethasone ophthalmic suspension every 4 hours. The moxifloxacin was reduced to every 4 hours again in alternating fashion with the 0.3% tobramycin/0.1% dexamethasone. The cyclopentolate was discontinued.
Five days later, the patient reported substantially better comfort and improved vision in the right eye. He continued to use the moxifloxacin and tobramycin/dexamethasone as instructed.
The corneal epithelial defect was only 0.5 mm in size, and the infiltrates appeared flatter and smaller. The immune ring was considerably less dense. There was no anterior chamber reaction noted. Of concern, there was a small symblepharon noted at the lateral canthus, right eye. The cultures continued to show no growth of bacterial or fungal colonies and the polymerase chain reaction assay result was negative. The moxifloxacin and tobramycin/dexamethasone were reduced to 3 times a day, alternating right eye. He was scheduled for a 2-week follow-up visit.
The patient presented with mild complaints of itching but increased comfort overall in both eyes. He had reduced the moxifloxacin and tobramycin/dexamethasone to twice a day, alternating every 6 hours. He restarted the tacrolimus 0.03% ointment around the eyelids in both eyes. He was using preservative-free artificial tears as needed in both eyes.
Biomicroscopy showed a closed corneal defect with a remnant infiltrate. The immune ring had dissipated. The bulbar conjunctiva had trace chemosis and injection. The overall appearance of the left eye was unchanged with persistent large palpebral papillae on the upper lids. There was minimal excoriation noted of the periocular skin. The intraocular pressure was 18 mm Hg in the right eye and 15 mm Hg in the left eye. There was no bacterial or fungal growth in the final culture assessment.
The moxifloxacin and tobramycin/dexamethasone were discontinued. The patient was placed on fluoromethalone 0.1% once a day for maintenance in the right eye for 2 weeks. The patient was instructed to use olopatadine 0.7% once a day in both eyes and artificial lubricants consistently. He was to continue to use the tacrolimus 0.03% ointment every other day on the periocular skin. He was scheduled for a 6-week follow-up examination.
The patient presented for the 6-week follow-up examination with minimal itching and irritation symptoms. He reported compliance with his medications and was using tacrolimus 0.03% ointment twice a day and olopatadine 0.7% once a day in both eyes. He uses cool compresses as needed and preservative-free artificial tears.
The examination showed uncorrected visual acuities of 20/25 in the right eye and 20/25 in the left eye. Slit lamp examination demonstrated lessening of the papillary response with minimal inferior corneal staining. The eyelid erosions had healed, and there was less edema and scaling. The intraocular pressure was 14 mm Hg in the right eye and 15 mm Hg in the left eye. There were no changes to the lens or posterior segment structures.
The patient was instructed to continue use of the tacrolimus 0.03% ointment once a day to eyelids and olopatadine 0.7% once a day, both eyes. Supportive treatment with lid hygiene, cool compresses, and artificial lubrication was emphasized. Compliance with the medication schedule was stressed. He was scheduled for a 6-month comprehensive visit.
DISCUSSION
Dupilumab is a fully human monoclonal immunoglobulin G antibody that blocks the shared interleukin 4 and interleukin 13 receptor, thus antagonizing the effects of these atopic mediators.12 Specific to atopic dermatitis, interleukin 4 and interleukin 13 are involved in barrier function and the development of xerosis, pruritus, and lichenification.1 In moderate to severe atopic dermatitis, systemic immunosuppression is necessary. Commonly, cyclosporine, steroids, and methotrexate are used to improve quality of life. Alternatively, biologics offer an advantage over these traditional therapies in atopic dermatitis through clearer skin, less pruritis, better sleep, and less side effects.1 Dupilumab received US approval for the treatment of atopic dermatitis in 2017. It is also approved for moderate to severe asthma, prurigo nodularis, and its efficacy has been shown in chronic rhinosinusitis with nasal polyps and eosinophilic esophagitis.13
The most common dupilumab-associated ocular surface disease findings includes blepharitis and tarsal inflammation. About 25% of patients had limbal inflammation.14 A review of the literature through June 2021 found the incidence of ocular surface disease associated with dupilumab varied from 8% to 26%.3 These early studies did not evaluate for preexisting ocular surface disease prior to starting dupilumab.3 A newer prospective study did evaluate for preexisting disease and found dupilumab-associated ocular surface disease developed in 28.9% of participants and was the most common adverse reaction associated with dupilumab in the treatment of atopic dermatitis.14 Onset of dupilumab-associated ocular surface disease occurs at 2 to 16 weeks, plateaus at 20 to 24 weeks, and is rare after 44 weeks following the start of dupilumab.12,15
Although corneal ulcers are known to occur with atopic eye disease, there are limited reports on corneal ulceration believed related to dupilumab-associated ocular surface disease.7–11 The cases associated with dupilumab vary in presentation and clinical signs. Two presented as bilateral peripheral ulcerative keratitis, two as paracentral defects that progressed to perforation, and another as a perilimbal infiltrate with corneal thinning. The pathogenesis has been speculated as either progressive atopic disease exacerbated by the dupilumab or an inflammatory reaction from resident flora enterotoxin.
None of the cases reported showed culture-positive infectious pathogens. Although this case presented similar to an infectious keratitis, the negative culture results and course with treatment does in part support a link to the use of dupilumab. Although the patient did not comply with the previous management, this patient also did not have the severe and rapid atopic keratoconjunctivitis changes, such as the increase in papillae, symblepharon, and the corneal ulceration, seen prior to beginning the dupilumab. This case is also consistent with the reported timelines of development of corneal ulceration that was reported to be between 2 and 12 weeks after beginning dupilumab. All cases, including this one, had between 1 and 2 dosages prior to onset of dupilumab-associated ocular surface disease.
Corneal ulceration is uncommon in atopic keratoconjunctivitis as well, with a retrospective study showing and incidence rate of 1.26%.7 The vast majority were linked to infectious pathogens, including Staphylococcus aureus or Streptococcus species.16–18 Atopic blepharitis has also been reported to be associated with increased risk of developing herpes simplex virus keratitis.19
Dupilumab-associated ocular surface disease is unique to atopic dermatitis for reasons unknown and not in the other Food and Drug Administration–approved uses. A review of multiple randomized clinical trials evaluating adverse side effects of dupilumab in the treatment of atopic conditions showed a higher incidence of conjunctivitis in atopic dermatitis compared with asthma and nasal polyposis groups.12 A proposed hypothesis is the inhibition of interleukin 4 results in a shift from T-helper cell 2- to T-helper cell 1–mediated inflammatory response, which exacerbates the chronic allergic T-helper cell 1 ocular inflammation associated with atopic dermatitis, as shown by the T-helper cell 1-associated elevated interferon-γ in tears.15
The pathogenesis of dupilumab-associated ocular surface disease is unknown but under investigation. Various hypotheses exist. Theories include a possible preexisting disorder, that is atopic keratoconjunctivitis, or a barrier dysfunction unique to atopic dermatitis and less common in other atopic diseases.12 Studies support that dupilumab-associated ocular surface disease may be a result of a reduction in goblet cells and mucin production.3 Interleukin 13 has been observed to increase goblet cell density and induce goblet cell differentiation, and its inhibition may cause goblet cell dysfunction and altered mucin production.1 Consistent with this theory is the higher risk of conjunctivitis with other interleukin 13 monoclonal antibodies, tralokinumab and lebrikizumab, used in atopic dermatitis treatment.1 Additionally, dupilumab-associated ocular surface disease was noted to improve when a Janus kinase inhibitor, ruxolitinib, was substituted for dupilumab.14 In a recent study, dupilumab did not reduce the overall number of goblet cells in dupilumab-associated ocular surface disease, but it did reduce the secretion of mucin5AC. Mucin5AC is the main gel-forming glycoprotein produced by goblet cells.14 This study also found lower median goblet cell numbers in both patients with more severe baseline atopic dermatitis and more severe dupilumab-associated ocular surface disease, suggesting low goblet cell density related to more severe ocular surface disease overall in atopic dermatitis.
The development of antidrug antibodies is another consideration based on research showing a higher incidence of ocular side effects that inversely correlates with lower serum levels of dupilumab and lower drug efficacy.1 However, findings in the SOLO-CONTINUE study do not support antidrug antibodies as the cause of dupilumab-associated ocular surface disease.20,21 The study also evaluated antidrug antibody levels across various treatment dosages and intervals. Results demonstrated an inverse correlation between treatment-emergent atopic dermatitis incidence and both dose and treatment interval with dupilumab, yet rate of conjunctivitis was low for all groups.20
Additional potential mechanisms suggest dupilumab-associated ocular surface disease pathophysiology is multifactorial. These include Malassezia colonization causing T-helper cell 17–driven dermatitis similar to seborrheic dermatitis, T-helper cell 1/T-helper cell 17 polarization, Demodex proliferation, and increased OX40 ligand activity.15 Some suggest dupilumab-associated ocular surface disease results from exacerbation of underlying allergic disease. However, biopsies of dupilumab-associated ocular surface disease patients did not show histological features of allergy, suggesting dupilumab-associated ocular surface disease is a separate entity from atopic keratoconjunctivitis.15
Treatment of atopic keratoconjunctivitis includes artificial tears, cool compresses, and lid hygiene. Topical ophthalmic mast cell stabilizers and topical antihistamines provide relief for itch. Topical immunosuppression, and systemic for severe cases, is necessary to address the inflammation.
In early cases of dupilumab-associated ocular surface disease, clinicians often advocated discontinuation of dupilumab. Newer evidence indicates dupilumab-associated ocular surface disease resolves with continuation of dupilumab in 80% of cases.12 Early treatment with anti-inflammatory medications minimizes the development of dupilumab-associated ocular surface disease. Tacrolimus ointment for external eyelids is frequently used, and a lower incidence of dupilumab-associated ocular surface disease is seen with use of anti-inflammatory treatment compared with nontreated or topical antihistamine use alone.14 There are no standard guidelines for the management of dupilumab-associated ocular surface disease, but a proposed algorithm includes a baseline eye examination prior to initiation of dupilumab, patch testing, and regular surveillance.15 However, cost-effectiveness and inconvenience of multiple visits may limit the practicality of this approach.
A different biologic, tralokinumab-ldrm (Adbry, LEO Phama, Inc) was approved in 2021 for the treatment of moderate to severe atopic dermatitis in pediatric patients aged 12 to 17 years. Like dupilumab, tralokinumab is associated with an increased occurrence of conjunctivitis. A study showed that the incidence was 7.5%, with most cases being mild or moderate. A majority of the cases resolved within the 16-week period of the trial. Increased risk of conjunctivitis occurred in patients who had more severe atopic dermatitis and an history of atopic keratoconjunctivitis.22
CONCLUSION
Mechanisms in which dupilumab causes or exacerbates ocular surface disease are yet to be determined. Corneal ulceration, whether infectious or immune related, is a rare but possible complication that may be associated with the use of dupilumab. To distinguish between atopic keratoconjunctivitis and dupilumab-associated ocular surface disease, baseline evaluation prior to initiation of dupilumab would be necessary. Most cases allow for continuation of dupilumab with topical treatment with anti-inflammatories, mast-cell stabilizers, and preservative-free artificial lubricants. Fortunately, most cases of dupilumab-associated ocular surface disease are mild, manageable, and temporary.
TAKE HOME POINTS
-
Dupilumab increases the risk of ocular surface disease in patients with atopic dermatitis.
-
Early recognition and treatment with topical corticosteroids or mast cell stabilizers is essential to decrease the development of long-term complications.
-
Corneal ulceration is rare but must be distinguished between infectious vs inflammatory causes.
-
It is critical to establish communication with dermatology to monitor for ocular signs while receiving therapy.
DISCLOSURES
None of the authors have a financial interest in the medications or products discussed in the manuscript.